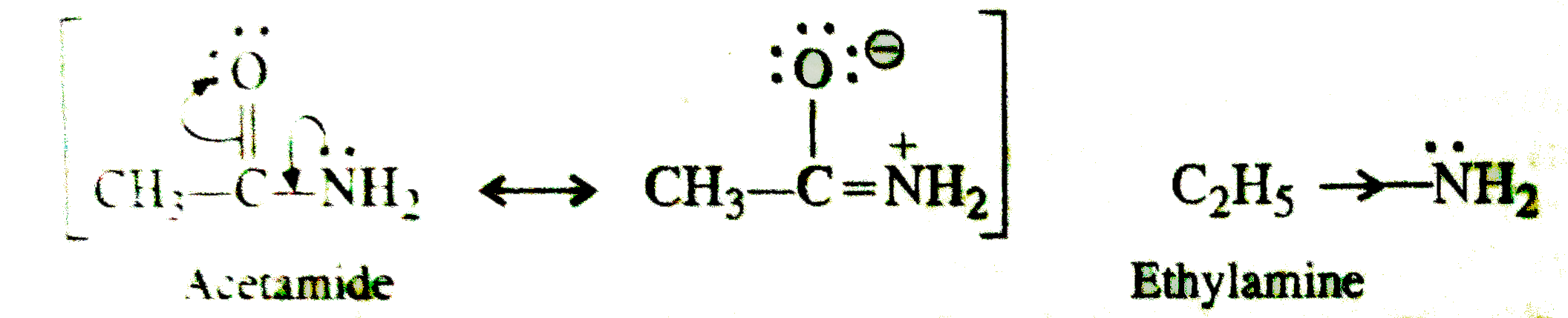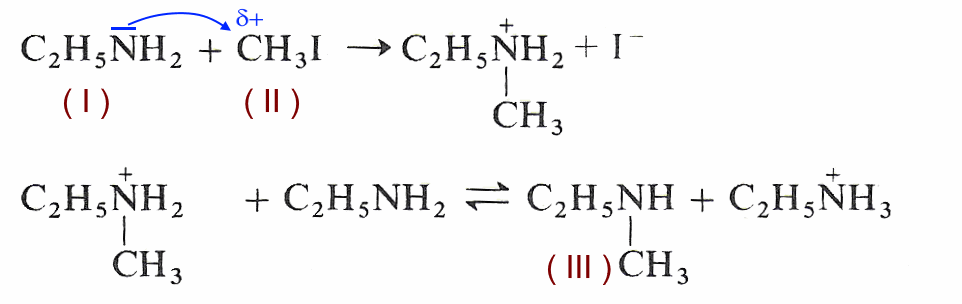
Ethylamine organic base molecule. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles: hydrogen (white), carbon (grey), nitrogen (blue Stock Photo - Alamy

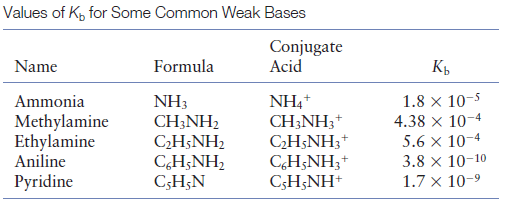
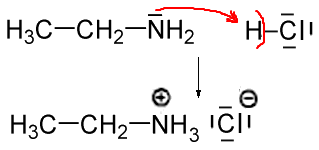
Acid-base chemistry of aliphatic amines weak bases pKb Kb values why stronger than aomatic amines reactions with acids primary secondary tertiary balanced neutralisation equations organic nitrogen compounds organonitrogen molecules advanced A level

Relative Basicity of Ammonia, Ethylamine & Phenylamine (7.6.3) | CIE A Level Chemistry Revision Notes 2019 | Save My Exams

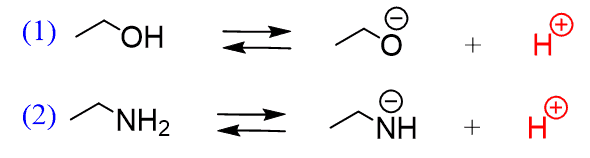
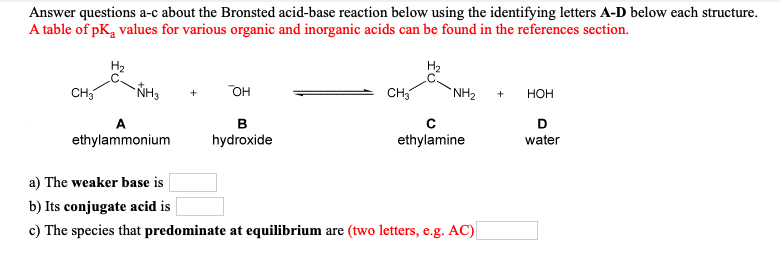
SOLVED:Which member of each pair is the stronger base? a. ethylamine or aniline b. ethylamine or ethoxide ion c. phenolate ion or ethoxide ion d. phenolate ion or acetate ion

Ethylamine organic base molecule. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles: hydrogen (beige), carbon (grey), nitrogen (blue Stock Photo - Alamy

Acid-base chemistry of aliphatic amines weak bases pKb Kb values why stronger than aomatic amines reactions with acids primary secondary tertiary balanced neutralisation equations organic nitrogen compounds organonitrogen molecules advanced A level
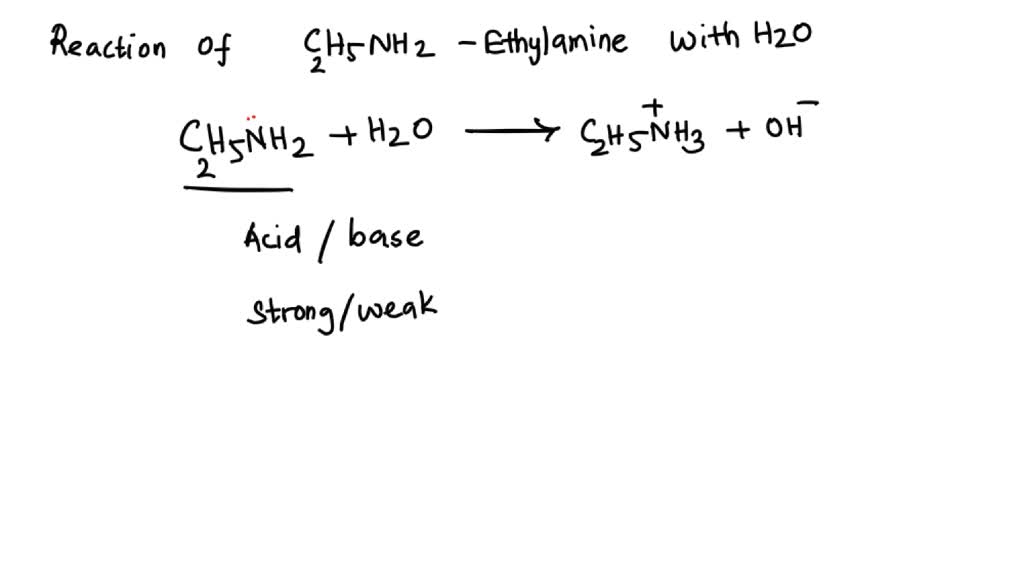
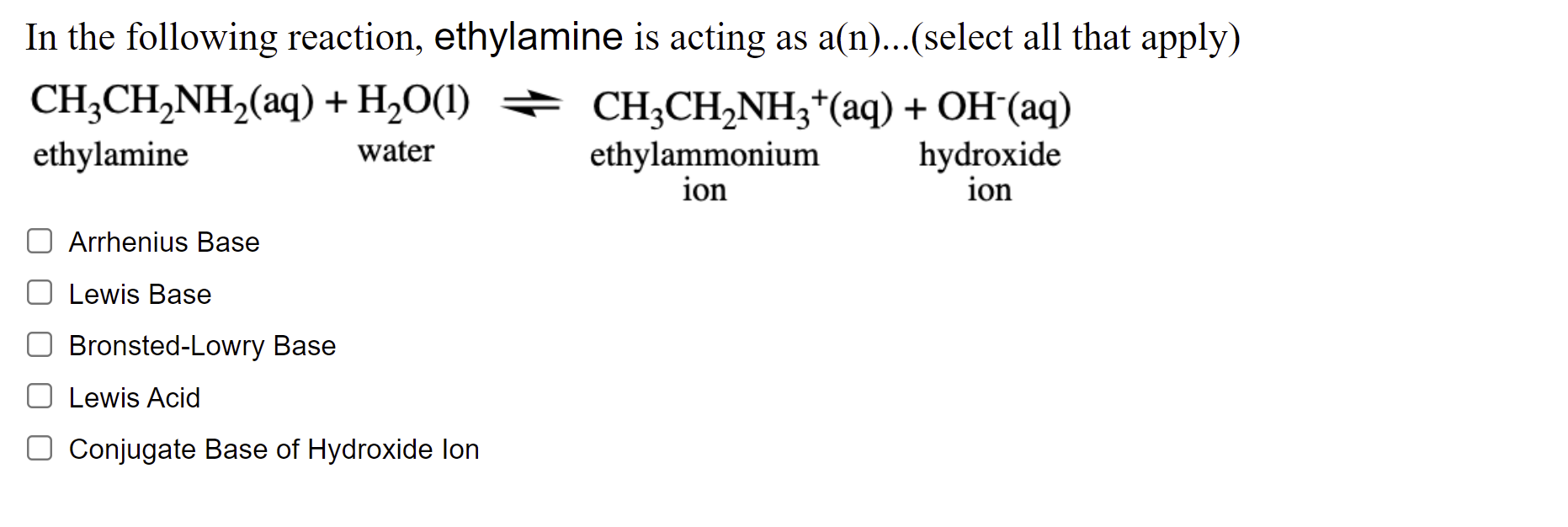
SOLVED: 5. When added to water, ethylamine undergoes the following reaction: C2H5NH2 + H2O C2H5NH3+ + OH- Is ethylamine an acid or a base in this reaction? Strong or weak? How do